by Erowid
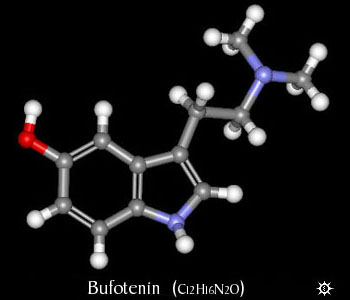
| NAME : | Bufotenin |
| CHEMICAL NAME : | 3-[2-(Dimethylamino)thyl]-1H-indol-5-ol |
| ALTERNATE CHEMICAL NAMES : | 3-(2-dimethylaminoethyl)-5-indolol; 5-hydroxy-N,N-dimethyltryptamine |
| ALTERNATE CHEMICAL NAMES : | N,N-dimethylserotonin, 3-(ß-dimethylaminoethyl)-5-hydroxyindole, mappine |
| CHEMICAL FORMULA | C12H16N2O |
| MOLECULAR WEIGHT | 204.27 |
| MELTING POINT | 174.5-178°C (Merck); 124-126°C (Ott) |
| From the Merck Index 12th Edition | |
|---|---|
MERCK INDEX ENTRY
1502. Bufotenine. 3-[2-(Dimethylamino)ethyl]-1H-indol-5-ol; 3-(2-dimethylaminoethyl)-5-indolol; 5-hydroxy-N,N-dimethyltryptamine; N,N-dimethylserotonin; 3-(Beta-dibethylaminoethyl)-5-hydroxyindole; mappine. C12H16N2O; mol wt 204.27. C 70.56%, H 7.89%, N 13.71%, O 7.83%. Isoln from toadds: Wieland et al., Ann. 513, 1 (1934); Weiland, Wieland, ibid. 528, 239 (1937); from toadstools: Wieland, Motzel, ibid. 581, 10 (1953). Isoln from Piptadenia peregrina Benth., Leguminosae: Stromberg, J. Am. Chem Soc. 76, 1707 (1954). Synthesis: Hoshino, Shimodaira, Ann. 520, 19 (1935; Harley-Mason, Jackson, Chem. & Ind. (London) 1952, 954; see also Serotonin; and Speetor, U.S. pat. 2,708,197 (1955 to Upjohn); Stoll et al., Helv. Chim Acta 38, 1452 (1955). Activity: Bhattacharya, Sanyal, Indian J. Physiol. Pharmacol. 15, 133 (1971). Crystal and molecular structure: G. Falkenberg, Acta Crystallogr. 28B, 3219 (1972).
Stout prisms from ethyl acetate, mp 146-147° bp0.1 320° uv max: 220, 265 nm (log E 4.0, 3.7). Almost insol in water. Freely sol in alcohol, less sol in ether. Sol in dil acids and alkalies.
Methyliodide, C13H19IN2O, stout prisms from methanol, dec 214-215°.
Note: This is a controlled substances (hallucinogen) listed in the U.S. Code of Federal Regulations, Title 21 Part 1308.11 (1995).