#54 2,5-DMA
DMA; 2,5-DIMETHOXYAMPHETAMINE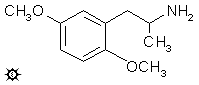
|
| [3D .mol structure] |
A solution of 17.0 g of 1-(2,5-dimethoxyphenyl)-2-nitropropene was prepared in 500 mL anhydrous Et2O. This solution was added slowly to a well-stirred suspension of 12.0 g LAH in 700 mL anhydrous Et2O. The mixture was then brought up to a reflux and maintained there for 20 h, cooled with an external ice bath, and the excess hydride destroyed by the cautious addition of H2O. Finally, a total of 500 mL H2O was added, followed by the addition of 300 g potassium sodium tartrate, and sufficient aqueous NaOH to bring the pH above 9. The two phases were separated, and the ether phase dried by the addition of anhydrous MgSO4. The drying agent was removed by filtration, and the clear filtrate saturated with a stream of anhydrous HCl gas. The formed crystals of 2,5-dimethoxyamphetamine hydrochloride (2,5-DMA) were removed by filtration, washed with anhydrous Et2O, and dried to constant weight of 16.3 g. Recrystallization from EtOH gave an analytical sample with a mp of 114-116 °C. The hydrobromide salt is reported to melt at 129-131 °C.
DOSAGE: 80 - 160 mg.
DURATION: 6 - 8 h.
EXTENSIONS AND COMMENTARY: The qualitative information on 2,5-DMA is very sparse. I was up to a 1+ with 80 milligrams of the hydrochloride, and since it appeared to be totally a physical trip with tremors and some cardiovascular push and nothing of a sensory nature, I chose to explore it no further. A report from South America found the intoxication to be largely pleasant (this, at 75 milligrams), with an enhanced interest in one's surroundings, but no perceptual changes, no overt stimulation, and no gross physiological effects other than a slight mydriasis (dilation of the pupils). I have also been told of a single trial of 250 milligrams of the tartrate (this is equivalent to somewhere in the 150-200 milligram range of the hydrochloride salt, depending upon the acid/base ratio of the tartrate salt) with some "speedy" effects but still no sensory changes. A seizure of capsules reported by the drug law enforcement authorities some 20 years ago found that each contained some 200 milligrams of the hydrobromide salt. This is equivalent to 170 milligrams of the hydrochloride salt, and suggests that level may be an effective dosage.
An intriguing, but little studied, analogue of 2,5-DMA is the compound with methyls in place of the methoxyls. 2,5-Dimethylamphetamine has been looked at, in man, as a potential anorexic, but there is little effect even at 150 milligrams. The 3,4-isomer, 3,4-dimethylamphetamine or xylopropamine, is an adrenergic agent and it has been found to be an analgesic in man at as little as 10 milligrams. This was assayed, rather remarkably, by attaching electrodes to the tooth fillings of the experimental subjects. But with this base, cardiovascular effects were not observed until doses of about 100 milligrams were administered, and toxic effects (nausea and vomiting) were reported at 150 milligrams. There was no suggestion of anything psychedelic.
All three isomers of monomethylamphetamine have also been looked at in man. The ortho- and meta-isomers, 2-methyl- (and 3-methyl- ) amphetamine are weak anorexics. At doses of up to 150 milligrams orally, there were signs of stimulation noted--talkativeness and loss of appetite. The para-isomer, 4-methyl-amphetamine or Aptrol, is more potent. At 75 milligrams (orally, in man) there is clear adrenergic stimulation, and at twice this dosage there are signs of mild toxicity such as salivation, coughing and vomiting.
There is a mystery, at least to me, concerning the commercial production of 2,5-DMA. At regular intervals, there is a public announcement of the production quotas that are requested or allowed by the Drug Enforcement Administration, for drugs that have been placed in Schedules I or II. In the Schedule I category there are usually listed amounts such as a gram of this, and a few grams of that. These are probably for analytical purposes, since there are no medical uses, by definition, for drugs in this Schedule. But there is a staggering quantity of 2,5-DMA requested, regularly. Quantities in the many tens of millions of grams, quantities that vie with medical mainstays such as codeine and morphine. I have heard that this material is used in the photographic industry, but I have no facts. Somewhere I am sure that there is someone who has to keep a lot of very careful books!
In the area of psychedelic drugs, the value of 2,5-DMA is mainly in its role as a precursor to the preparation of materials that can come from a direct electrophilic attack on the activated 4-position. These uses can be found under things such as DOB and DOI and DON. The radio-halogenation of N-substituted homologues of 2,5-DMA with hypoiodite or hypofluorite is part of an extensive study underway in the search for radio-labeled brain blood flow agents. The rationale for this work is to be found in the commentary under IDNNA. In essence it has been found that the N-substitution or N,N-disubstitution of 2,5-DMA where the 4-position is unsubstituted and thus available for the introduction of a radioactive nucleus can give rise to potentially useful drugs. Most of these 2,5-dimethoxy exploratory compounds were made by the reductive alkylation of 2,5-dimethoxy-4-(radio)iodophenylacetone, using various mono or dialkyl amines. This, too, is described under IDNNA.
However, the study of various direct iodinations and fluoridations that would have the N,N-dimethyl substitution on the amphetamine nitrogen atom, would require the 4-proteo- analogue, and this was made from the above nitrostyrene. A solution of the above nitrostyrene, 22.3 g 1-(2,5-dimethoxyphenyl)-2-nitropropene in 100 mL acetic acid was added to a suspension of elemental iron in acetic acid (45 g in 250 mL) and worked up with water and base washing to give, after distillation at 92-106 °C at 0.35 mm/Hg, 13.8 g 2,5-dimethoxyphenylacetone as a pale yellow oil. This underwent reductive amination with dimethylamine hydrochloride in MeOH solution, using sodium cyanoborohydride, to give the target compound 2,5-dimethoxy-N,N-dimethylamphetamine oxalate with a melting point of 133-134 °C (4.6 g ketone gave 1.38 g of salt). Anal. (C15H23NO6) C,H. It has also been prepared by the N,N-dimethylation of 2,5-DMA directly, with formaldehyde and formic acid. This has been called 2,5-DNNA, or IDNNA without the "I." This intermediate, 2,5-DNNA, underwent direct radioiodination with labeled iodine monochloride in the presence of perchloric acid to give IDNNA with a 40% incorporation of isotope. Reaction with labeled acetyl hypofluorite, on the other hand, gave only a 2% in-corporation of the radio-isotope. This latter compound is, chemically, 4-fluoro-2,5-dimethoxy-N,N-dimethylamphetamine and, using the reasoning suggested above and with IDNNA, might best be encoded FDNNA.
The 2,5-dimethylamphetamine analogue mentioned above was also explored in this IDNNA concept. The commercially available 2,5-dimethylbenzaldehyde was converted to the nitrostyrene with nitroethane (1-(2,5-dimethylphenyl)-2-nitropropene, yellow crystals with a melting point of 24.5-25.5 °C) which reacted with elemental iron in acetic acid to give the ketone 2,5-dimethylphenylacetone (boiling at 140-150 °C at 0.4 mm/Hg). Reductive amination with dimethylamine and sodium cyanoborohydride gave 2,5-DMNNA (2,5,N,N-tetramethylamphetamine) as a clear oil with a boiling point of 115-125 °C at 0.35 mm/Hg. It gave poor yields of the 4-fluoro analogue with acetyl hypofluorite.
All of these latter materials remain unevaluated in man.
| [ |
[Main Index] | [Forward |