#73 EEE
2,4,5-TRIETHOXYAMPHETAMINE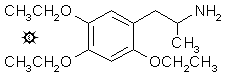
|
| [3D .mol structure] |
To a mixture of 10.5 g N-methyl formanilide and 11.9 g POCl3 that had incubated at room temperature for 0.5 h (it had become quite red in color) there was added 6.4 g of the solid ether, 1,2,4-triethoxybenzene. The mixture was heated on the steam bath for 2.5 h, then poured into 500 mL of shaved ice. After a few minutes stirring, crystals appeared. The reaction was allowed to stand for a few h, then filtered and sucked as dry as possible. The damp 14.4 g of slate-green crude solids were dissolved in 30 mL boiling MeOH, and allowed to cool to room temperature overnight. Filtration of the cream-colored product, and air drying, gave 6.1 g of 2,4,5-triethoxybenzaldehyde with a mp of 94-95 °C. A solution containing 0.5 g of this aldehyde and 0.4 g malononitrile in 7 mL absolute EtOH was treated with three drops of triethylamine. There was an immediate formation of granular yellow crystals of 2,4,5-triethoxybenzalmalononitrile which, on filtering and air drying, weighed 0.4 g and had a mp of 169-170 °C.
A solution of 5.0 g 2,4,5-triethoxybenzaldehyde and 2.6 g nitroethane in 14.8 g glacial acetic acid was treated with 1.6 g anhydrous ammonium acetate and heated on the steam bath for 2 h. The addition of an equal volume of H2O gave a slightly turbid solution which, upon the administration of a small amount of externally developed seed, smoothly set up as orange crystals as the reaction mix returned to room temperature. The product was removed by filtration, washed with a little 50% acetic acid, and allowed to air dry to constant weight. There was thus obtained 2.5 g of fluffy yellow-orange (almost yellow) crystals of 2-nitro-1-(2,4,5-triethoxyphenyl)propene with a mp of 91-92.5 °C. Anal. (C15H21NO5) C,H.
To a gently refluxing suspension of 1.7 g LAH in 200 mL anhydrous Et2O under a He atmosphere, there was added 2.5 g 2-nitro-1-(2,4,5-triethoxyphenyl)propene by allowing the condensing Et2O to drip into a shunted Soxhlet thimble containing the nitrostyrene, thus effectively adding a warm saturated solution of the nitrostyrene dropwise. Refluxing was maintained for 5 h, and then the reaction mixture was cooled with an external ice bath. The excess hydride was destroyed by the cautious addition of 300 mL 1.5 N H2SO4. When the aqueous and Et2O layers were finally clear, they were separated, and 50 g of potassium sodium tartrate were dissolved in the aqueous fraction. Aqueous NaOH was then added until the pH was above 9, and this was extracted with 3x200 mL CH2Cl2. Removal of the solvent under vacuum produced an amber oil that was dissolved in anhydrous Et2O and saturated with anhydrous HCl gas. After a few min delay, there com-menced the separation of fine white crystals of 2,4,5-triethoxyamphetamine hydro-chloride, (EEE). These weighed, after filtration, Et2O washing, and air drying to constant weight, 1.75 g and had a mp of 167-168 °C, with prior softening at 162 °C. Anal. (C15H26ClNO3) C,H,N.
DOSAGE: unknown.
DURATION: unknown.
EXTENSIONS AND COMMENTARY: This amphetamine, the final item on the ethoxy homologue of TMA-2 project, has never been tried in man. I do not know how it tastes, but I suspect that it is probably bitter. An interesting sidelight concerning this project, and one which can serve as a measure of the enthusiasm that went into it, is that (except for the 2-ethoxy homologue EMM) all of the possible ethoxy homologues of TMA-2, including MEM, MME, EEM, EME, MEE and EEE, their precursor nitrostyrenes, the precursor aldehydes (and their malononitrile derivatives), the precursor ethers, and the precursor phenols, for a total of 33 compounds, were all synthesized, purified, and characterized within a period of just over three weeks. Actually it was 23 days, and that was a magically exciting time.
And there were two true treasures that came out of it all. The compound MEM, and the knowledge that the 4-position was where the action is.
| [ |
[Main Index] | [Forward |